|
Forest sites
|
Type of site
|
Bird
species
|
Bird family
|
Migration
status
|
Conservation
status
|
Food
habits
|
|
Golfito
|
Harvested/ High
crop intensity
|
Attila spadiceus
Catharus ustulatus
|
Tyrannidae
Turdidae
|
Resident
Migratory
|
Low concern
|
Insects, spiders,
small frogs, lizards, berries and seeds
Fruits, seeds, some insects and
invertebrates
|
|
Santa Helena
|
Non-harvested/
High crop intensity
|
Dendrocincla fuliginosa
|
Furnariidae
|
Resident
|
Low concern
|
Insects and spiders
|
|
Chacha-lacas
|
Non-harvested/
Low crop intensity
|
Glyphorhynchus
spirurus
Thamnophilus atrinucha
Phaenostictus mcleannani
|
Furnariidae
Thamnophilidae
Thamnophilidae
|
Residents
|
ND
Low concern
Low concern
|
Insects and spiders
Insects, spiders, and lizards
Insects, spiders, scorpions, other
invertebrates and small vertebrates
|
Note: Mixed generalized linear model results are included as
supplementary data.
DISCUSSION
Habitat loss and fragmentation are among the
primary causes of biodiversity loss, increasing the risk of species extinction (Fahrig,
2003). Forest harvesting has been related to changes in the components and
attributes of biodiversity (Putz
et al., 2001), which are conditioned by factors such as the ecological
requirements of the species (Woltmann,
2000), the intensity of harvesting (Mason,
1996), and logging schemes and techniques (Bawa
& Seidler, 1998).
In Costa Rica, a selective harvesting scheme has
been implemented, and legislation establishes a minimum of 15 years between
harvests. One study in northern Costa Rica forest fragments found that the
estimated richness of birds and number of individuals per trophic guild showed
no significant differences between harvested (16 years previous to the study)
and non-harvested forests (Sánchez
Núñez, 2009). However, these parameters tended to be higher in non-harvested
forests. Nevertheless, species composition varied between forests:
non-harvested forests had more species of hummingbirds, nuthatches, antbirds,
and psittacine than harvested forests. Species occurrence for all trophic
guilds differed between non-harvested and harvested forests, with this parameter
tending to be greater in non-harvested forests. Sánchez
Núñez (2009) stated that these harvested forests seemed to be recovering after
harvesting, although more research was needed to ensure sufficient time for a
total recovery of bird communities after forest harvesting. Similarities found
in this study in the number of bird species, total number of individuals, and
Shannon diversity indices between harvest or fragmentation classes seem to
agree with those found by Sánchez
Núñez (2009).
Concerning parasite infection of birds in northern
Costa Rica, mites were the most common ectoparasites found in this study,
similar to previous reports (Enout
et al., 2012). However, we found a low percentage of chewing lice. In
Costa Rica, Rallicola ochrolaemi, R. fuliginosa, and R. cephalosa
were reported previously in bird species such as Automolus ochrolaemus, Dendrocincla
fuliginosa, and Glyphorynchus spirurus (Sychra
et al., 2007). On the other hand, we found a higher tick infestation rate
of birds (12.5%) than the 7% reported by other authors in different wild birds
in seven protected areas on the Caribbean and Pacific slopes of Costa Rica (Ogrzewalska
et al., 2015). A low percentage of coccidian parasites was found, while
other studies have reported Eimeria and Isospora as common and
occasional (Cole
& Friend, 1999). Time of day, age, and feeding habits affect coccidian
oocyst shedding in wild passerines, with more of these parasites found in the
afternoon (López
et al., 2007). However, these captures were carried out in the morning. It
is also possible that physiological differences in feeding habits between
species could have affected these results (Boughton,
1933; Dolnik, 1999a, 1999b). Additionally, gastrointestinal parasites are not
excreted constantly, and the use of a single fecal sample per bird may have
limited the detection of these parasites; serial sampling would have been ideal
but was not feasible under the conditions of this field study.
Six ticks (Amblyomma spp.), found in six
different birds, were positive for Rickettsia amblyommatis. These ticks
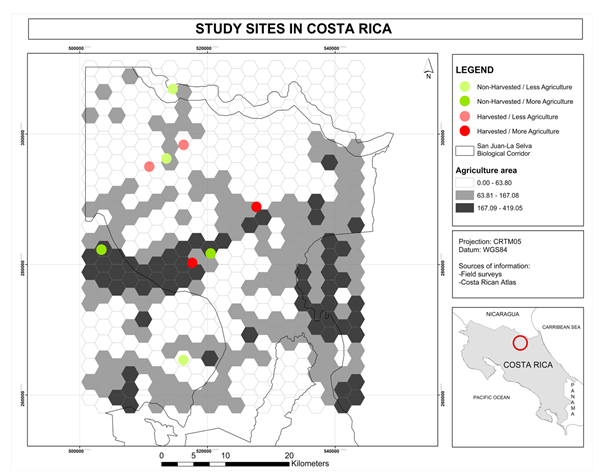
were found in three forest sites: two in forests surrounded by high crop
intensity areas and one in a forest surrounded by low crop intensity areas.
Other researchers have proposed that a higher prevalence of ectoparasites (in
this case, ticks) on birds in fragmented areas of Brazil might be caused by the
dominance of a few generalist bird species in small patches, allowing easier
transmission of parasites among individuals (Ogrzewalska
et al., 2011). In addition, in more fragmented areas, we might expect a
more heterogeneous landscape, with forest fragments interspersed with other
classes of land use, for example, pastures for cattle or other productive uses.
This might, in turn, maximize contact not only among individuals of wildlife
species in forest fragments but even between wildlife and domestic animals
living in the matrix surrounding forest fragments. This scenario may increase
access to cattle and cattle ticks (Tolesano-Pascoli et al., 2010), which
is relevant considering all avian species with Rickettsia spp. positive
ticks usually visit the ground to seek food or collect material to build nests (Sick,
1997).
A non-expected pattern was found in the case of
ectoparasite infection and Rickettsia spp. positives in areas with or
without forest harvesting; two of the three forest sites with ticks positive
for Rickettsia spp. were found in non-harvested sites and one in a
harvested site (harvested at least 15 years before sampling). It might be
expected that birds inhabiting forests subjected to disturbances by logging
would be more stressed or have lower immune defenses (Klukowski
& Nelson, 2001; Tieleman et al., 2005), which might, in turn, affect
their parasite load and lead to increased prevalence of a disease agent in the
environment (Deem
et al., 2001; Patz et al., 2000).
In addition, according to the dilution effect, we
might expect a higher proportion of parasites in more disturbed and possibly
less diverse forests, such as harvested areas (Ostfeld
& Keesing, 2000). However, a higher proportion of Proctophyllodes
sp. in non-harvested forests was found. Moreover, a higher diversity of birds
was found in the harvested forest than in the non-harvested forest, although
this difference was not statistically significant. However, the intermediate
disturbance hypothesis may be considered here – a mechanism proposed by Connell
(1978), to explain the maintenance of diversity in tropical forests and coral
reefs. This hypothesis states that intermediate disturbance in mature
ecosystems maintains higher levels of species richness and biodiversity than in
the absence of such disturbances (Roxburgh
et al., 2004). The similarity of bird richness and diversity among forest
sites, and the absence of disease agents in birds found in this study, may
account for a tendency towards recovery taking place in bird communities of
harvested forests from northern Costa Rica, given the selective forest
harvesting scheme and the period between harvests (16 years) applied to these
forests. However, it is necessary to identify consequences in the birds' health
when forests are harvested and in the first years following that harvesting.
The other infectious agents (Anaplasma spp.
and Borrelia spp.) analyzed in ticks were negative in this study. In
Europe, Borrelia burgdorferi s.l. was found and transmitted, especially
by Ixodes ricinus ticks (Gern
et al., 1998), which are reported in the highlands of Costa Rica (Ogrzewalska
et al., 2015). We also obtained negative results for chlamydiosis in
birds. Studies around the world on chlamydiosis in Passeriformes are not
common, mainly because they are not considered to play an important role in the
transmission of the bacteria (Kaleta
& Taday, 2003). However, a few studies have reported these species as
carriers of the bacterium (Olsen
et al., 1998; Rehn et al., 2013; Simpson & Bevan, 1989).
In general, the bird communities analyzed in this
study might be diverse enough to dilute the probability of the presence of most
of the infectious agents analyzed, consistent with the dilution effect (Ostfeld
& Keesing, 2000). These authors suggested that habitat loss and
fragmentation can contribute to disease outbreaks indirectly through the loss
of biodiversity while maintaining high biodiversity might decrease the
prevalence of an infectious disease among hosts due to the dilution effect. It
is also possible that the avian communities in this study were similar because
they were in a similar and relatively small landscape, probably shaped by the
same biophysical factors and human productive activities. Future studies that
consider a larger number of forest sites in areas showing much larger
differences in forest size and fragmentation may provide further insights into
the effects of habitat loss and fragmentation on disease ecology.
These results seem to support the dilution effect
proposed for the effects of habitat fragmentation on disease transmission.
However, the forest sites analyzed were similar in terms of health status and
the presence of disease agents in birds. This may reflect a possible tendency
towards recovery in the harvested forests studied or a strong similarity among
forest sites because of their closeness in a generally similar landscape.
ACKNOWLEDGMENTS
We thank Vicerrectoría de Investigación,
Universidad Nacional de Costa Rica, for the translation of this manuscript; Área
de Conservación Huetar Norte, that granted research permits for this study;
Angie Sánchez Núñez and Carol Sánchez Núñez, who significantly contributed to
data collection and analysis, and the support given by all the farm owners
who provided us access to their properties. This work was supported by the
Fondo de Educación Estatal Superior (FEES) from the Consejo Nacional de
Rectores de Costa Rica (CONARE).
ETHICS, CONFLICT OF INTEREST, AND
FUNDING STATEMENT
The authors declare that we have fully complied
with all pertinent ethical and legal requirements, both during the study and in
the production of the manuscript; that there are no conflicts of interest of
any kind; that all financial sources are fully and clearly stated in the
acknowledgements section; and that they fully agree with the final edited
version of the article. A signed document has been filed in the journal
archives.
The statement of each author’s contribution to the
manuscript is as follows: All co-authors: Study design. K.B.P., A.E.J.R., and
G.D.: Laboratory analysis. M.R.I.: Data collection and analysis. All
co-authors: preparation and final approval of the manuscript.
REFERENCES
Alberti, A., Addis, M. F., Sparagano, O., Zobba, R., Chessa, B.,
Cubeddu, T., Parpaglia, M. L. P., Ardu, M., & Pittau, M. (2005). Anaplasma
phagocytophilum, Sardinia, Italy. Emerging Infectious Diseases, 11(8),
1322–1324. https://doi.org/10.3201/eid1108.050085
Atyeo, W., & Braasch,
N. (1966). The Feather Mite Genus Proctophyllodes (Sarcoptiformes:
Proctopbyllodidae). Bulletin of the University of Nebraska State Museum. https://digitalcommons.unl.edu/museumbulletin/39
Bawa, K. S.,
& Seidler, R. (1998). Natural forest management and conservation of
biodiversity in tropical forests. Conservation biology, 12(1),
46–55.
Boughton, D. C. (1933).
DIURNAL GAMETIC PERIODICITY IN AVIAN ISOSPORA. American Journal of
Epidemiology, 18, 161–184.
http://aje.oxfordjournals.org/content/18/1/161.extract
Chassot, O. (2006). Biología de la conservacion de la Lapa Verde (1994-2006): 12 años de
experiencia = 12 years of conservation biology of the great green macaw 1994-
2006. Centro Cientifico Tropical.
Clay, T. (1953). Revisions
of the genera of Mallophaga—I. The Rallicola-complex. Proceedings of the
Zoological Society of London, 123, 563–587.
http://phthiraptera.info/sites/phthiraptera.info/files/38919.pdf
Cole, R. A., & Friend,
M. (1999). Parasites and Parisitic Diseases (Field Manual of Wildlife
Diseases). Other Publications in Zoonotics and Wildlife Disease, Paper
15, 188–258.
Connell, J. H. (1978).
Diversity in tropical rain forests and coral reefs. Science (New York, N.Y.),
199(4335), 1302–1310. https://doi.org/10.1126/science.199.4335.1302
Daszak, P., Cunningham, A.
A., & Hyatt, A. D. (2000). Emerging infectious diseases of wildlife–threats
to biodiversity and human health. science, 287(5452), 443–449. http://science.sciencemag.org/content/287/5452/443.short
Deem, S. L.,
Karesh, W. B., & Weisman, W. (2001). Putting Theory into Practice:
Wildlife Health in Conservation. Conservation Biology, 15(5),
1224–1233. https://doi.org/10.1111/j.1523-1739.2001.00336.x
Dobson, A. P., & May,
R. M. (1986). Disease and conservation. En M. E. Soulé (Ed.), Conservation
Biology: The Science of Scarcity and Diversity (pp. 345–365). Sinauer
Associates.
Dolnik, O. V. (1999a).
Diurnal oocyst periodicity in Isospora dilatata (Sporozoa, Eimeriidae) from the
common starling (Sturnus Vulgaris) in nature. Parasitologiya, 33(1),
74–80.
Dolnik, O. V. (1999b).
Diurnal periodicity in appearance of Isospora (Protozoa: Coccidea) oocysts from
some passerine birds. Proc. Zool. Inst. RAS, 281, 113–118.
Dolz, G.,
Castro, R., Jiménez-Rocha, A. E., Retamosa, M., & Alberti, A. (2019). Strain
diversity of Rickettsia amblyommatis in ticks infesting birds in the North
Huetar conservation area of Costa Rica. Ticks and Tick-Borne Diseases, 10(5),
1109–1112. https://doi.org/10.1016/j.ttbdis.2019.06.007
Enout, A. M. J., Lobato, D.
N. C., Diniz, F. C., & Antonini, Y. (2012). Chewing lice (Insecta,
Phthiraptera) and feather mites (Acari, Astigmata) associated with birds of the
Cerrado in Central Brazil. Parasitology Research, 111(4),
1731–1742. https://doi.org/10.1007/s00436-012-3016-5
ESRI. (2009). ArcGIS®9.3
Geocoding Technology (Versión 9.3) [Software]. ESRI. www.esri.com
Fahrig, L. (2003). Effects
of Habitat Fragmentation on Biodiversity. Annual Review of Ecology,
Evolution, and Systematics, 34(1), 487–515.
https://doi.org/10.1146/annurev.ecolsys.34.011802.132419
Fimbel, R.
A., Grajal, A., & Robinson, J. G. (Eds.). (2001). The Cutting
Edge: Conserving Wildlife in Logged Tropical Forests. Columbia University
Press; JSTOR. https://doi.org/10.7312/fimb11454
Gern, L., Estrada-Peña, A.,
Frandsen, F., Gray, J. S., Jaenson, T. G. T., Jongejan, F., Kahl, O.,
Korenberg, E., Mehl, R., & Nuttall, P. A. (1998). European Reservoir Hosts
of Borrelia burgdorferi sensu lato. Zentralblatt Für Bakteriologie: Medical
Microbiology, Virology, Parasitology, Infectious Diseases, 287(3),
196–204. https://doi.org/10.1016/S0934-8840(98)80121-7
Kaimowitz, D. (1996). Livestock
and Deforestation in Central America in the 1980s and 1990s: A Policy
Perspective. CIFOR.
Kaleta, E. F., & Taday,
E. M. A. (2003). Avian host range of Chlamydophila spp. Based on isolation,
antigen detection and serology. Avian Pathology: Journal of the W.V.P.A,
32(5), 435–461. https://doi.org/10.1080/03079450310001593613
Kaltenboeck,
B., Kousoulas, K. G., & Storz, J. (1991). Detection and strain
differentiation of Chlamydia psittaci mediated by a two-step polymerase chain
reaction. Journal of Clinical Microbiology, 29(9), 1969–1975.
King, K. C., Daniel
Mclaughlin, J., Boily, M., & Marcogliese, D. J. (2010). Effects of
agricultural landscape and pesticides on parasitism in native bullfrogs. Biological
Conservation, 143(2), 302–310.
https://doi.org/10.1016/j.biocon.2009.10.011
Klukowski, M., &
Nelson, C. E. (2001). Ectoparasite Loads in Free-Ranging Northern Fence
Lizards, Sceloporus undulatus hyacinthinus: Effects of Testosterone and Sex. Behavioral
Ecology and Sociobiology, 49(4), 289–295.
http://www.jstor.org/stable/4601889
Labruna, M. B., Whitworth,
T., Horta, M. C., Bouyer, D. H., McBride, J. W., Pinter, A., Popov, V.,
Gennari, S. M., & Walker, D. H. (2004). Rickettsia species infecting
Amblyomma cooperi ticks from an area in the state of São Paulo, Brazil, where
Brazilian spotted fever is endemic. Journal of Clinical Microbiology, 42(1),
90–98.
Larki, S., Alborzi, A.,
Chegini, R., & Amiri, R. (2018). A Preliminary Survey on Gastrointestinal
Parasites of Domestic Ducks in Ahvaz, Southwest Iran. Iranian
Journal of Parasitology, 13(1), 137–144.
López, G.,
Figuerola, J., & Soriguer, R. (2007). Time of day, age and feeding
habits influence coccidian oocyst shedding in wild passerines. International
Journal for Parasitology, 37(5), 559–564. https://doi.org/10.1016/j.ijpara.2006.12.014
Mason, D. (1996). Responses
of Venezuelan Understory Birds to Selective Logging, Enrichment Strips, and
Vine Cutting. Biotropica, 28(3), 296–309.
https://doi.org/10.2307/2389194
Mason, D. J., &
Thiollay, J. (2001). En R. A. Fimbel, A. Grajal, & J. G. Robinson (Eds.), The
Cutting Edge: Conserving Wildlife in Logged Tropical Forest. Columbia
University Press.
Massung, R. F., Slater, K.,
Owens, J. H., Nicholson, W. L., Mather, T. N., Solberg, V. B., & Olson, J.
G. (1998). Nested PCR Assay for Detection of Granulocytic Ehrlichiae. Journal
of Clinical Microbiology, 36(4), 1090–1095.
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC104695/
Mironov, S. V. (1985).
Feather mites of the genera Analges and Pteronyssoides from the European part
of the USSR (Sarcoptiformes, Analgoidea)]. Parazitologicheskii
Sbornik, 33, 159–208.
Nava, S.,
Venzal, J. M., González-Acuña, D., Martins, T. F., & Guglielmone, A. A.
(2017). Chapter 4—Morphological Keys for Genera and Species of Ixodidae
and Argasidae. En S. Nava, J. M. Venzal, D. González-Acuña, T. F. Martins,
& A. A. Guglielmone (Eds.), Ticks of the Southern Cone of America
(pp. 323–336). Academic Press.
https://doi.org/10.1016/B978-0-12-811075-1.00004-2
Ogrzewalska, M., Literák,
I., Capek, M., Sychra, O., Calderón, V. Á., Rodríguez, B. C., Prudencio, C.,
Martins, T. F., & Labruna, M. B. (2015). Bacteria of the genus Rickettsia
in ticks (Acari: Ixodidae) collected from birds in Costa Rica. Ticks and
Tick-Borne Diseases, 6(4), 478–482.
https://doi.org/10.1016/j.ttbdis.2015.03.016
Ogrzewalska, M., Uezu, A.,
Jenkins, C. N., & Labruna, M. B. (2011). Effect of Forest Fragmentation on
Tick Infestations of Birds and Tick Infection Rates by Rickettsia in the
Atlantic Forest of Brazil. EcoHealth, 8(3), 320–331.
https://doi.org/10.1007/s10393-011-0726-6
Olsen, B., Persson, K.,
& Broholm, K. A. (1998). PCR detection of Chlamydia psittaci in faecal
samples from passerine birds in Sweden. Epidemiology and Infection, 121(2),
481–484. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2809549/
Orwig, K. (1968). The
Genera and Species of the Feather Mite Subfamily Trouessartinae except Trouessartia
(Acarina: Proctophyllodidae). Bulletin of the University of Nebraska State
Museum, 8, 1–179. https://digitalcommons.unl.edu/museumbulletin/110
Ostfeld, R.
S., & Keesing, F. (2000). Biodiversity and Disease Risk: The Case of
Lyme Disease. Conservation Biology, 14(3), 722–728.
Park, C., & Atyeo, W.
(1971). A Generic Revision of the Pterodedinae, a New Subfamily of Feather
Mites (Sarcoptiformes: Analgoidea). Bulletin of the University of Nebraska
State Museum, 9(3), 39–88.
https://digitalcommons.unl.edu/museumbulletin/108
Patz, J. A.,
Graczyk, T. K., Geller, N., & Vittor, A. Y. (2000). Effects of
environmental change on emerging parasitic diseases. International Journal
for Parasitology, 30(12–13), 1395–1405.
Ministerio
de Agricultura y Ganadería (1998). Principios,
criterios e indicadores de manejo forestal y certificación
(Publicación No. 27388-MINAE, N° 212). Recuperado de http://www.mag.go.cr/legislacion/1998/de-27388.pdf
Putz, F.
E., Blate, G. M., Redford, K. H., Fimbel, R., & Robinson, J. (2001). Tropical
Forest Management and Conservation of Biodiversity: An Overview. Conservation
Biology, 15(1), 7–20.
R Core Team. (2013). R:
A language and environment for statistical computing. R Foundation for
Statistical Computing. http://www.R-project.org/
Regnery, R. L., Spruill, C.
L., & Plikaytis, B. D. (1991). Genotypic identification of rickettsiae and
estimation of intraspecies sequence divergence for portions of two rickettsial
genes. Journal of Bacteriology, 173(5),
1576–1589. http://jb.asm.org/content/173/5/1576
Rehn, M.,
Ringberg, H., Runehagen, A., Herrmann, B., Olsen, B., Petersson, A. C.,
Hjertqvist, M., Kühlmann-Berenzon, S., & Wallensten, A. (2013). Unusual
increase of psittacosis in southern Sweden linked to wild bird exposure,
January to April 2013. Euro Surveillance: Bulletin Européen Sur Les Maladies
Transmissibles = European Communicable Disease Bulletin, 18(19),
20478.
Rijpkema, S.
G., Molkenboer, M. J., Schouls, L. M., Jongejan, F., & Schellekens, J. F.
(1995). Simultaneous detection and genotyping of three genomic groups of
Borrelia burgdorferi sensu lato in Dutch Ixodes ricinus ticks by
characterization of the amplified intergenic spacer region between 5S and 23S
rRNA genes. Journal of Clinical Microbiology, 33(12), 3091–3095.
Roxburgh, S. H., Shea, K.,
& Wilson, J. B. (2004). The Intermediate Disturbance Hypothesis: Patch
Dynamics and Mechanisms of Species Coexistence. Ecology, 85(2), 359–371.
Sánchez
Núñez, A. (2009). Ensamble y ocurrencia de aves en bosques sometidos a
manejo forestal, Región Huetar Norte, Costa Rica. Tesis
-- Universidad Nacional, Heredia.
Sick, H.
(1997). Ornitologia Brasileira—Helmut Sick 2ed-01. Editora Nova
Fronteira.
https://es.scribd.com/doc/100102495/Ornitologia-Brasileira-Helmut-Sick-2ed-01
Simpson, V. R., &
Bevan, R. (1989). Chlamydia psittaci infection in robins. The Veterinary
Record, 125(21), 537.
Sychra, O., Literák, I.,
Capek, M., & Havlícek, M. (2007). Chewing Lice (Phthiraptera) From
Ovenbirds, Leaftossers and Woodcreepers (Passeriformes: Furnariidae:
Furnariinae, Sclerurinae, Dendrocolaptinae) from Costa Rica, With Descriptions
of Four New Species of the Genera Rallicola and Myrsidea. Caribbean Journal
of Science, 43(1), 117–126. https://doi.org/10.18475/cjos.v43i1.a11
Tenter, A. M., Barta, J.
R., Beveridge, I., Duszynski, D. W., Mehlhorn, H., Morrison, D. A., Andrew
Thompson, R. C., & Conrad, P. A. (2002). The conceptual basis for a new
classification of the coccidia. ASP Special, 32(5), 595–616.
https://doi.org/10.1016/S0020-7519(02)00021-8
Tieleman, B. I., Williams,
J. B., Ricklefs, R. E., & Klasing, K. C. (2005). Constitutive innate
immunity is a component of the pace-of-life syndrome in tropical birds. Proceedings
of the Royal Society of London B: Biological Sciences, 272(1573),
1715–1720. https://doi.org/10.1098/rspb.2005.3155
Woltmann,
S. (2000). Comunidades de aves del bosque en áreas alteradas y no alteradas de
la concesión forestal La Chonta, Santa Cruz, Bolivia. Documento
Técnico, 92, 2000.
Yamaouti, S.
(1961). Systema Helnrinthum. Vol. III. The Nematodes of Vertebrates. Systema
Helnrinthum. Vol. III. The Nematodes of Vertebrates., Part II,
681–1261. CABI Databases.
Zhou, S., Chen, D., Cai,
W., Luo, L., Low, M., Tian, F., Tay, V., Ong, D., & Hamilton, B. (2010).
Crowd modeling and simulation technologies. ACM Transactions on Modeling and
Computer Simulation (TOMACS), 20(4), 1–35.
https://doi.org/10.1145/1842722.1842725
![]() , Ana Eugenia Jiménez-Rocha2
, Ana Eugenia Jiménez-Rocha2![]() , Gaby Dolz3
, Gaby Dolz3![]() , Mónica Retamosa-Izaguirre4
, Mónica Retamosa-Izaguirre4![]()